A web module produced by Committee 3 of the International Commission on Radiological Protection (ICRP)
What is the purpose of this document?
In the past 100 years, diagnostic radiology, nuclear medicine and radiation therapy have evolved from the original crude practices to advanced techniques that form an essential tool for all branches and specialties of medicine. The inherent properties of ionising radiation provide many benefits but also may cause potential harm.
In the practice of medicine, there must be a judgement made concerning the benefit/risk ratio. This requires not only knowledge of medicine but also of the radiation risks. This document is designed to provide basic information on radiation mechanisms, the dose from various medical radiation sources, the magnitude and type of risk, as well as answers to commonly asked questions (e.g radiation and pregnancy). As a matter of ease in reading, the text is in a question and answer format.
Interventional cardiologists, radiologists, orthopaedic and vascular surgeons and others, who actually operate medical x-ray equipment or use radiation sources, should possess more information on proper technique and dose management than is contained here. However, this text may provide a useful starting point.
The most common ionising radiations used in medicine are X, gamma, beta rays and electrons. Ionising radiation is only one part of the electromagnetic spectrum. There are numerous other radiations (e.g. visible light, infrared waves, high frequency and radiofrequency electromagnetic waves) that do not posses the ability to ionize atoms of the absorbing matter. The present text deals only with the use of ionising radiation in medicine.
Is the use of ionising radiation in medicine beneficial to human health?
Yes. Benefit to patients from medical uses of radiation has been established beyond doubt.
Modern diagnostic radiology assures faster, more precise diagnosis and enables monitoring of a large proportion of diseases. It has been estimated that in about one half of cases, radiological procedures (plain film radiography, fluoroscopy, computed tomography) have a substantial impact on the speed of diagnosis and in a large fraction of cases they are of decisive importance. Furthermore, several screening procedures (such as mammography) have been developed which are beneficial for specific populations at relatively high risk of some diseases. In addition a number of interventional radiological procedures (e.g angioplasty), introduced in the last 10-20 years, contribute significantly to the effectiveness of treatment of very serious and life threatening diseases of the cardiovascular, central nervous system and other organ systems. These procedures are also cost-effective.
Nuclear medicine uses radioactive substances, called radiopharmaceuticals, in the diagnosis and treatment of a range of diseases. These substances are especially developed to be taken up predominantly by one organ, or type of cell in the body. Following their introduction into the body for diagnostic purposes they are followed either by external measurements, yielding images of their distribution (both in space and in time), or by activity measurements in blood, urine and other media. In all cases the information obtained is of functional character. This information is not obtainable, or obtainable with less accuracy, by other modalities. Nuclear medicine offers therefore unique diagnostic information in oncology (diagnosis and staging), cardiology, endocrinology, neurology, nephrology, urology and others. Most of the methods currently in use are those of choice in the diagnostic process, because they show high sensitivity, specificity and good reproducibility. Their cost-effectiveness is also high. In addition it should be emphasized that these procedures are non-invasive and present no risk of direct complications to the patient.
One has to remember that whereas electrical generators of ionising radiation (X-ray machines, electron accelerators) stop emitting radiation when switched off, radioactive sources do emit radiation, which cannot be modified in course of the radioactive decay. This means that some precautions may have to be taken with such patients given large therapeutic amounts of radionuclides when they are in a hospital and afterwards when they go home -to protect against exposure of the staff, relatives, friends and members of the public.
Radiation therapy uses ionising radiation for treatment. The incidence of cancer is about 40%, reflecting long life expectancy. Cancer also leads to ~ 20-30 per cent cumulative mortality. Current medical practice uses radiotherapy in about 1/2 of newly diagnosed cancer cases. Therapeutic techniques can be highly complex, and place very high demands on the accuracy of irradiation. To be effective they must be approached on an interdisciplinary basis, requiring effective and harmonious cooperation between radiation oncologists, medical physicists and highly qualified technicians. However, it should be remembered that radiotherapy of cancer is often accompanied by adverse side effects of the treatment. Some adverse effects are unavoidable and often resolve spontaneously or with treatment. Serious adverse effects may occur and result from the proximity of sensitive normal tissues to the treatment field or rarely as a result of individual radiation sensitivity. They do not undermine the purpose of radiotherapy. Appropriate use of radiotherapy saves millions of lives every year overall. Even if only palliative treatment is possible the therapy substantially reduces suffering. There are also some non-malignant diseases whose treatment by radiation is a method of choice.
Radiotherapy using radiopharmaceuticals is generally non-invasive but limited to several well-established situations where killing hyperfunctioning or malignant cells is important (for example hyperthyroidism, cancer of the thyroid, degenerative and inflammatory diseases of joints, palliative treatment of metastases to the skeleton). In addition, there are many studies showing significant potential for radio-labelled antibodies and receptor -avid peptides to be used in the treatment of several malignancies. However, this mode of treatment is still in its early days.
Ionising radiation is, therefore, one of the basic tools of contemporary medicine, both in diagnosis and therapy. Practice of contemporary, advanced medicine, without use of ionizing radiation appears currently unthinkable.
Are there risks to the use of ionising radiation in medicine?
There obviously are some risks. The magnitude of risk from radiation is dose-related with higher amounts of radiation being associated with higher risks. The undisputed health benefits of diagnostic X-ray and nuclear medicine diagnostics may be accompanied by a generally small risk (probability) of deleterious effects. This fact has to be taken into account while using ionising radiation sources in diagnosis. Since large amounts of radiation are required in radiation therapy, the risk of radiation-related adverse effects is measurably higher.
The aim of managing radiation exposure is to minimise the putative risk without sacrificing, or unduly limiting, the obvious benefits in the prevention, diagnosis and also in effective cure of diseases (optimisation). It should be pointed out that when too little radiation is used for diagnosis or therapy there is an increase in risk although these risks are not due to adverse radiation effects per se. Too low an amount of radiation in diagnosis will result in either an image that does not have enough information to make a diagnosis and in radiation therapy, not delivering enough radiation will result in increased mortality because the cancer being treated will not be cured.
Experience has provided ample evidence that reasonable selection of conditions, under which ionising radiation is being used in medicine, can lead to health benefits substantially outweighing the estimated possible deleterious effects.
How do we quantify the amount of radiation?
The frequency or intensity of biological effects is dependant upon the total energy of radiation absorbed (in joules) per unit mass (in kg) of a sensitive tissues or organs. This quantity is called absorbed dose and is expressed in gray (Gy). Some X or gamma rays will pass through the body without any interaction, and they will produce no biological effect. On the other hand, the radiation which is absorbed, may produce effects. Absorbed doses of radiation can be measured and/or calculated and they form basis for evaluation of the probability of radiation-induced effects.
In evaluating biological effects of radiation after partial exposure of the body further factors such as the varying sensitivity of different tissues and absorbed doses to different organs have to be taken into consideration. To compare risks of partial and whole body irradiation at doses experienced in diagnostic radiology and nuclear medicine a quantity called effective dose is used. It is expressed in sievert (Sv). Effective dose is not applicable to radiation therapy, where very large absorbed doses affect individual tissues or organs.
What do we know about the nature (mechanism) of radiation-induced biological effects ?
Cells can be killed by radiation During cellular division chromosomal aberrations due to radiation may result in loss of part of the chromosomal DNA which results in cell death. The probability of chromosomal aberrations is proportional to dose and those cells free of critical damage to DNA retain their dividing potential.
Surviving cells may carry changes in the DNA at a molecular level (mutations). Elementary, primary damage to DNA results from chemical damage by free radicals, originating from radiolysis of water. DNA damage also can result from the direct interaction of ionising particles with the DNA double helix (rarely).
Important changes in DNA occur in the form of breaks in continuity of the DNA chains although other forms of damage also arise. These breaks may affect one strand of the helix (single strand breaks, SSB) or both strands in the same location (double strand breaks, DSB). SSB occur very frequently in the DNA without irradiation, and are easily and effectively repaired by specific enzyme systems. In contrast, many induced DSB are more complicated and less easily repaired. As a result, a significant proportion of the damage is repaired incorrectly (mis-repair). These mis-repaired breaks can lead to chromosomal aberrations and gene mutations. Some of the genes mutated in such a way form the first step (initiation) of the very long and complicated process of carcinogenesis, requiring also several subsequent mutations (most likely not induced by radiation) in the affected cells. Similar mutation mechanisms, when affecting germinative cells, may lead to hereditary mutations expressed in descendants of the irradiated persons. Of course, the essential point in considering these possible sequelae of irradiation is the frequency (or probability of occurrence) of undesired effects in persons irradiated with a given dose, or in their descendants.
How are effects of radiation classified?
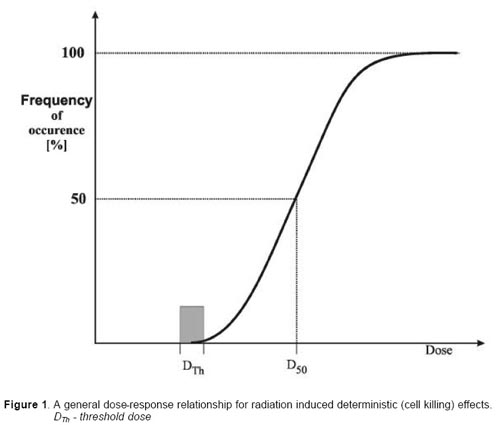
There are two basic categories of the biological effects that may be observed in irradiated persons. These are 1) due largely to cell killing (deterministic) and 2 mutations which may result in cancer and hereditary effects (stochastic or probabilistic ). Effects due to cell killing (such as skin necrosis) have a practical threshold dose below which the effect is not evident but in general when the effect is present its severity increases with the radiation dose. The threshold dose is not an absolute number and vary somewhat by individual. Effects due to mutations (such as cancer) have a probability of occurrence that increases with dose, it is currently judged that there is not a threshold below which the effect will not occur and finally the severity of the effects is independent of the dose. Thus a cancer caused by a small amount of radiation can be just as malignant as one caused by a high dose.
Deterministic effects. These effects are observed after large absorbed doses of radiation and are mainly a consequence of radiation induced cellular death. They occur only if a large proportion of cells in an irradiated tissue have been killed by radiation, and the loss cannot be compensated by increased cellular proliferation. The ensuing tissue loss is further complicated by inflammatory processes and, if the damage is sufficiently extensive, also by secondary phenomena at the systemic level (e.g. fever, dehydration, bacteraemia etc.). In addition, eventual effects of healing processes, e.g. fibrosis, may contribute to additional damage and loss of function of a tissue or an organ. Clinical examples of such effects are: necrotic changes in skin, necrosis and fibrotic changes in internal organs, acute radiation sickness after whole body irradiation, cataract, and sterility (table 1).
Doses required to produce deterministic changes are in most cases large (usually in excess of 1-2 Gy). Some of those occur in a small proportion of patients as side effects of radiotherapy. They can also be found after complex interventional investigations (such as vascular stenting) when long fluoroscopy times have been used.
The relationship between the frequency of a given deterministic effect and the absorbed dose has a general form presented in fig. 1. It can be seen that the essential feature of this dose-response relationship is the presence of a threshold dose. Below this dose no effect may be diagnosed but with increasing dose the intensity of the induced damage increases markedly, in some situations, dramatically. An example of the deterministic damage to the skin is presented in fig. 2.
Malformations induced by radiation in the conceptus in the period of organogenesis (3-8 week of pregnancy), are also due to cell killing and are classified as deterministic effects. The same applies to malformations of the forebrain -leading to mental retardation -induced by the exposure between 8 and 15 weeks (and to some extent up to 25 week) after conception. The threshold doses are however, substantially lower than those found for deterministic effects after irradiation in extrauterine life: thus, 100-200 mGy form a threshold-range for malformations induced between the 3d and 8th week, and ~ 200 mGy for the aforementioned brain damage (8-25 week).
Stochastic effects. As mentioned above, irradiated and surviving cells may become modified by induced mutations (somatic, hereditary). These modifications may lead to two clinically significant effects: malignant neoplasms (cancer) and hereditary mutations.
Cancer: Ionising radiation is a carcinogen although a relatively weak one. Careful follow-up of over 80,000 atom bomb survivors in Hiroshima and Nagasaki over the last 50 years indicates that there have been 12,000 cancer cases of which less than 700 excess deaths were due to radiation. Expressed another way, only about 6% of the cancer occurring in these survivors is radiation-related.
These observations allow estimation of the probability with which a given dose may lead to diagnosis (incidence) and death (mortality) from various cancers. Among the latter there are several forms of leukaemia and solid tumours of different organs, mostly carcinomas of the lung, thyroid, breast, skin and gastrointestinal tract. Radiation-induced cancers do not appear immediately after radiation exposure but require time to become clinically apparent (latent period). Examples of minimum latent periods are non-CLL leukemia 2 years, about 5 years for thyroid and bone cancer and 10 years for most other cancers. Mean latent periods are 7 years for non-CLL leukemia and more than 20 years for most other cancers. It is important to note that some tumours do not appear to be radiation-induced or only weakly so. These include carcinomas of the prostate, cervix, uterus, lymphomas and chronic lymphatic leukemia.
Hereditary effects: The risk of hereditary effects of ionising radiation has been estimated on basis of experiments on various animal species, because there are no demonstrated effects in humans (the likely values of probability per unit dose are given later).
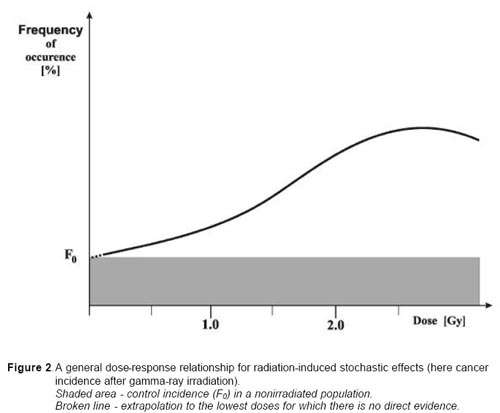
From careful analysis of the experimental studies and epidemiological surveys it may be concluded that dose-response relationships for these two categories of stochastic effects have a distinctly different form from those characterising deterministic sequelae. A general dose-response relationship for cancer is presented in fig. 3. The principal features of the relationship may be summarised as follows: not be interpreted as the presence of a dose threshold. It is assumed that at the low doses (< 0.2 Gy), probability of the effect (frequency) increases most likely proportionally with the dose.
| a.b. | The induction of cancer by X or gamma rays yields increasing frequency of the effect with increasing dose up to a maximum above which the curve flattens off, with possible decline at still higher doses. At the lower end of the curve, below ~ 100-200 mGy, any potential effect cannot be measured easily because statistical errors of the observations due to the large amount of spontaneous cancer and the impact of confounding factors. This should |
| 4 |
c. There is always a spontaneous frequency of the effect (mutations, cancer) in non-irradiated populations (F0 in Fig 2), which cannot be differentiated qualitatively from that induced by radiation. In fact, mutations or cancers induced by irradiation have the same morphological, biochemical, and clinical etc. characteristics as the cases occurring in non-irradiated individuals
What is magnitude of the risk for cancer and hereditary effects?
1. Analysis of the epidemiological data of irradiated populations has allowed derivation of the approximate risk of radiation-induced cancer. The lifetime value for the average person is roughly a 5% increase in fatal cancer after a whole body dose of 1 Sv (which is much higher than would found in most medical procedures). A statistically significant increase in cancer has not been detected in populations exposed to doses of less than 0.05 Sv.
It appears that the risk in fetal life, in children and adolescents exceeds somewhat this average level (by a factor of 2 or 3) and in persons above the age of 60 it should be lower roughly by a factor of ~ 5 (due to limited life expectancy and therefore less time available for manifestation of a cancer, which is a late appearing effect of the exposure).
The higher dose diagnostic medical procedures (such a CT scan of the abdomen or pelvis) yield an effective dose of about 10 mSv. If there were a large population in which every person had 1 such scan, the theoretical lifetime risk of radiation induced fatal cancer would be about 1 in 2,000 (0.05%). This can be compared to the normal spontaneous risk of fatal cancer which is about 1 in 4 (25%).
Individual risk may vary from theoretical calculations. The cumulative radiation dose from medical procedures is very small in many individuals, however in some patients the cumulative doses exceed 50 mSv and the cancer risk should be carefully considered. Many relatively high dose diagnostic procedures (such as CT) should be clearly justified and when this is done, benefit will far outweigh risk. Unjustified procedures at any dose level should be avoided. In radiotherapy there is a risk of second cancers but the risk is small compared to the imperative to treat the current malignancy.
Hereditary effects as a consequence of radiation exposure have not been observed in humans. No hereditary effects have been found in studies of the offspring and grandchildren of the atomic bomb survivors. However, as based on animal models and knowledge of human genetics, the risk of hereditary deleterious effects have been estimated to not be greater than 10% of the radiation induced carcinogenic risk.
Is ionizing radiation from medical sources the only one to which people are exposed?
No. All living organisms on this planet, including humans, are exposed to radiation from natural sources. An average yearly effective dose from this so-called natural background, amounts to about 2.5 mSv. This exposure varies substantially geographically (from 1.5 to several tens of mSv in limited geographical areas). Artificial sources - except medicine - add very minute doses to population at large.
What are typical doses from medical diagnostic procedures?
Various diagnostic radiology and nuclear medicine procedures cover a wide dose range based upon the procedure. Doses can be expressed either as absorbed dose to a single tissue or as effective dose to the entire body which facilitates comparison of doses to other radiation sources (such as natural background radiation. Typical values of effective dose for some procedures are presented in Table 2. The doses are a function of a number of factors such as tissue composition, density and thickness of the body. For example, it takes less radiation to penetrate the air in the lungs for a chest radiograph than to penetrate the tissues of the abdomen.
One should also be aware that even for a given procedure there may be a wide variation in the dose given cfor the same procedure on a specific individual when performed at different facilities. This variation may be up to a factor of ten and is often due to differences in the technical factors for the procedure such as film/screen speed, film processing, and voltage. IN addition there often are even wider variations in and among facilities for a given type of procedure due to less than satisfactory conduct of the procedure in some facilities.
Can radiation doses in diagnosis be managed without affecting the diagnostic benefit?
Yes. There are several ways to reduce the risks to very, very low levels while obtaining very beneficial health effects of radiological procedures, far exceeding the health impact of a possible detriment. In this context it should be mentioned also that high ratio of benefit vs. radiological risk depends to large extent on a good methodology of procedures and high quality of their performance. Therefore quality assurance and quality control in diagnostic radiology and nuclear medicine play also a fundamental role in the provision of appropriate, sound radiological protection of the patient.
There are several ways that will minimise the risk without sacrificing the valuable information that can be obtained for patients' benefit. Among the possible measures it is necessary to justify the examination before referring a patient to the radiologist or nuclear medicine physician.
Repetition should be avoided of investigations made recently at another clinic or hospital. Results of the investigations should be recorded in sufficient detail in patients' documentation, and carried over to another health-care unit. This rule could result in avoidance of a significant fraction of unnecessary examinations.
Failure to provide adequate clinical information at referral may result in a wrong procedure or technique being chosen by radiologist or nuclear medicine specialist. The result may be a useless test, with the investigation contributing only to patients' exposure.
An investigation may be seen as a useful one if its outcome - positive or negative - influences management of the patient. Another factor, which potentially adds to usefulness of the investigation, is strengthening confidence in the diagnosis.
To fulfill these criteria, indications for specific investigation both in the general clinical situation, and in a given individual patient, must be made by the referring physician on basis of medical knowledge. Difficulties may arise in the referral procedure mainly due to the dynamic development of the field of medical imaging. Technical progress in medical radiology and nuclear medicine has been enormous over the last 30 years; in addition two new modalities have entered the field: ultrasound and magnetic resonance imaging. It is not surprising therefore, that following the technical developments may be difficult both to a general practitioner and even specialists in many fields of medicine. There are, however, several published guidelines (see suggested readings), which may help in making an appropriate referral, using well-founded criteria, based on clinical experience and epidemiology.
The most important circumstances that should be taken into account to avoid inappropriate referrals can be broadly categorised as follows: possibility of obtaining similar information without using ionising radiation, i.e. by means of ultrasound (US) or magnetic resonance imaging (MRI). Their use is indicated where these modalities are available, and when the cost (this applies mostly to MRI), waiting times and organisational difficulties are not prohibitive. The guidelines mentioned above provide also information when these modalities are preferable as a starting and sometimes the only investigation to be performed.
Are there situations when diagnostic radiological investigations should be avoided?
Yes. There are well-established views -not always respected - which indicate that in some circumstances radiography or fluoroscopy does not contribute anything to patients' management. This applies to situations when a disease could not have progressed or resolved since the previous investigation, or the data obtained could not influence patients' treatment.
Most common examples of unjustified examinations include: routine chest radiography at admission to a hospital or before surgery in absence of symptoms indicating cardiac or pulmonary involvement (or insufficiency); skull radiography in asymptomatic subjects of accidents; lower sacro-lumbal radiography in stable degenerative condition of the spine in the 5th or later decade of life, but there are of course many others.
Screening of asymptomatic patients for presence of a disease may be done only if national health authorities made a decision that high incidence in a given age bracket, high efficacy of early disease detection, low exposure of screened individuals, and easily available and effective treatment may result in high benefit vs. risk ratio. Positive examples include fluoroscopy or radiography for detection of tuberculosis in societies or groups with high prevalence of the disease, mammography for early detection of breast cancer in women after 50 y of age, or screening for gastric carcinoma by specialised contrast fluoroscopy in countries with high incidence of this disease. All factors involved in screening must be periodically reviewed and reassessed. If positive criteria cease to be satisfied the screening should be discontinued.
Irradiation for legal reasons and for purposes of insurance should be carefully limited or excluded. Generally irradiation of individuals for legal reasons is without medical benefit. One of common examples is that insurance companies may require various X-ray investigations to satisfy the expectation that a person to be insured is in good health. In numerous cases such demands, particularly in asymptomatic individuals, should be treated with caution and often appear unjustified when they are medically not in the direct interest of the person concerned.
Are there special diagnostic procedures that should have special justification?
While all medical uses of radiation should be justified, it stands to reason that the higher the dose and risk of a procedure, the more the medical practitioner should consider whether there is a greater benefit to be obtained. There are radiological procedures that deliver doses at the upper end of the scale, presented in table 2.
Among these special position is occupied by computed tomography (CT), and particularly its most advanced variants like spiral or multi slice CT. Usefulness and efficacy of this great technical achievement is beyond doubt in particular clinical situations, however the ease of obtaining results by this mode and temptation to monitor frequently the course of a disease or perform screening should be tempered by the fact that repeated examinations may deliver an effective dose of the order of 100 mSv, a dose for which there is direct epidemiological evidence of carcinogenicity.
Do children and pregnant women require special consideration in diagnostic procedures?
Yes. Both the fetus and children are thought to be more radiosensitive than adults. Diagnostic radiology and diagnostic nuclear medicine procedures (even in combination) are extremely unlikely to result in doses that cause malformations or a decrease in intellectual function. The main issue following in-utero or childhood exposure at typical diagnostic levels (<50 mGy) is cancer induction.
Before a diagnostic procedure is performed it should be determined whether a patient is, or may be, pregnant, whether the fetus is in the primary radiation area and whether the procedure is relatively high dose (e.g. barium enema or pelvic CT scan). Medically indicated diagnostic studies remote from the fetus (e.g. radiographs of the chest or extremities, ventilation/perfusion lung scan) can be safely done at any time of pregnancy if the equipment is in proper working order. Commonly the risk of not making the diagnosis is greater than the radiation risk.
If an examination is typically at the high end of the diagnostic dose range and the fetus is in or near the radiation beam or source, care should be taken to minimize the dose to the fetus while still making the diagnosis. This can be done by tailoring the examination and examining each radiograph as it is taken until the diagnosis is achieved and then terminating the procedure. In nuclear medicine many radiopharmaceuticals are excreted by the urinary tract/ In these cases maternal hydration and encouraging voiding will reduce bladder residence time of the radiopharmaceutical and therefore will reduce fetal dose.
For children, dose reduction in achieved by using technical factors specific for children and not using routine adult factors. In diagnostic radiology care should be taken to minimize the radiation beam to only the area of interest. Because children are small, in nuclear medicine the use of administered activity lower than that used for an adult will still result in acceptable images and reduced dose to the child.
What can be done to reduce radiation risk during the performance of a diagnostic procedure?
The most powerful tool for minimising the risk is appropriate performance of the test and optimisation of radiological protection of the patient. These are the responsibility of the radiologist or nuclear medicine physician and medical physicist
The basic principle of patients' protection in radiological X-ray investigations and nuclear medicine diagnostics is that necessary diagnostic information of clinically satisfactory quality should be obtained at the expense of a dose as low as reasonably achievable, taking into account social and financial factors.
Evidence obtained in numerous countries indicates that the range of entrance doses (i.e. doses measured at surface of the body at the site where X-ray beam is entering the body) for a given type of radiographic examination is very wide. Sometimes the lowest and highest doses, measured in individual radiological installations, vary by a factor of ~ 100. As most measured doses tend to group at the lower end of the distribution (fig. 4 Need to add figure and legend of dose distribution for a specific examination) it is clear that the largest doses, above, for instance, 70-80 percentile of the distribution, cannot be reasonably justified. By establishing the so called diagnostic reference levels for each of principal investigations at such a percentile, one can identify the places (institutions, X-ray machines) in need of corrective actions, which will easily and substantially reduce the average dose to patients on a country-wide scale.
This goal may be reached by co-operation of radiologists with medical physicists and auditing persons or teams. There are numerous technical factors, which when systematically applied, reduce exposure significantly. The effort to optimise protection requires good organisation as well as permanent willingness and vigilance to keep the doses as low as reasonably achievable. It may be easily shown that the risk, even if it is quite small, can still be reduced several - fold compared with the situation prevailing in previous decades.
Among the procedures that should be avoided are: 1) fluoroscopy and photofluorography for screening for tuberculosis in children and adolescents (only normal radiographs should be made instead at this age). 2) Fluoroscopy without electronic image intensification. In most developed countries such procedure - which gives quite high doses to the patient - is now legally forbidden.
It should be emphasised, that radiological interventional procedures lead to higher doses to patients than normal diagnostic investigations. However, indications for such procedures in most cases result from a high risk from conventional surgery. Appropriate modern equipment and training of personnel allow the patients' exposure to be limited to an acceptable level, securing a very high benefit/risk ratio.
In nuclear medicine the magnitude of dose to the patient results principally from the activity1/ of the administered radiopharmaceutical. The range of activity of the latter, administered for a given purpose, varies among different departments by a small factor -usually a factor of three spans the highest and the lowest values. In several countries there are established respective reference or recommended levels and exceeding those should be usually avoided in examination of an individual of standard size. There are also accepted rules (formulae) for changing the activity as a function of body mass and for reducing the activity given to children relative to that administered to adults. Typical effective doses to patients in diagnostic nuclear medicine are in a similar range as those observed in X-ray diagnostics (Table 2). Good procedures and adherence to the principles of quality assurance and quality control secure a high benefit: risk ratio for the properly justified examinations. During pregnancy, investigations using radiopharmaceutical should be treated in a similar way as normal radiographic procedures. Accordingly, they should be performed only if no alternative diagnostic methods are available and if the investigations cannot be delayed until after delivery. To avoid serious damage to the foetal thyroid any procedure employing free 131I ions -even in small activities -is contraindicated starting with ~ 10-12 weeks of the pregnancy (when foetal thyroid becomes functional).
Lactating women may be investigated with radiopharmaceuticals. There are some radiopharmaceuticals that are relatively long lived and which are excreted in breast milk (such as iodine-131). After administration of such radiopharmaceuticals, breast-feeding must be discontinued to avoid transfer to the child. There are, however other radionuclides that are short lived (such as most technetium-99m compounds) that may not require discontinuation of breast-feeding or only for a few hours or a day.
1/ - Activity -number of nuclear disintegration per second (dps) in a given sample. Used as a measure of quantity of radioactive substances, here radiopharmaceuticals administered to patients. The unit is the becquerel which is 1 dps. A megabequerel (MBq) is 1 million dps.
The optimisation of patients' protection is based on a principle that the dose to the irradiated target (tumour) must be as high as it is necessary for effective treatment while protecting the healthy tissues to the maximum extent possible.
............................................................................................................................................................................................What can be done to reduce radiation risk during conduct of radiation therapy?
Radiotherapy based on proper indications is frequently a successful way of prolonging the life of a patient or of reducing suffering when only palliation is possible, thus improving the patient’ssible, thus improving the paties quality of life. To achieve this success requires the highest standards of performance (accuracy of delivered dose), both when planning irradiation for an individual patient and in actual delivery of the dose.
The decision to undertake a radiotherapy course is optimally made through a multidisciplinary team including surgeons, medical and radiation oncologists. This discussion should confirm the justification of the procedure, absence of more beneficial alternative treatments and commonly the optimal way of combining different techniques (radiotherapy, surgery and chemotherapy). When such a multidisiplinary approach is not possible, the radiation oncologist making the decision alone should keep in mind the alternative treatments or combine treatment strategies.
Actually, while the generic justification of radiotherapy cannot be questioned in the vast majority of cases. Increasing efforts are being made, in some cases, to decrease the delivered dose and to reduce the irradiated volumes. This is particularly true for some specific types of cancers, such as Hodgkin;s disease and for cancers of children, where the almost constant association with chemotherapy may allow the radiation oncologist to reduce dose and irradiated volume and a subsequent reduction in adverse side effects.
In a large number of cases, decreasing the dose to the target volume is not possible since it would unacceptably decrease the cure rate. In these cases present technological developments aim at optimising the patients protection, keeping the absorbed tumor dose as high as is necessary for effective treatment while protecting nearby healthy tissues. Conformal therapy has helped greatly in this regard.
It should be remembered that successful eradiaction of a malignant tumor by radiation therapy requires high-absorbed doses and there is a delayed (and usually low) risk of late complication. The above mention techniques are used to provide the best benefit/risk ratio.
Can pregnant women receive radiotherapy?
A malignant tumour in a pregnant woman may require radiotherapy in attempt to save life of the patient. If a tumour is located in a distant part of the body, the therapy - with individually tailored protection of the abdomen (screening) - may proceed. If the beam needs to be closer to the conceptus but still not irradiating the latter directly, special precautions need to be taken and an expert in dosimetry should make calculations of the dose to the fetus before the decision of starting the therapy is made. A dose to the conceptus (3-8 weeks post conception) from direct irradiation by the primary beam will reach values exceeding substantially thresholds for malformations of various organs, or of the brain (8 to 25 weeks) with resulting mental retardation in post-uterine life. It may also lead to stunting of foetal growth, even if the treatment took place in 3rd trimester of pregnancy.
It should be also remembered that irradiation of the fetus in all trimesters of the pregnancy carries an increased risk of cancer in the newborn in the first or second decade of life and at therapeutic doses -or their significant fraction -this risk can be substantial. Therefore, in view of all mentioned factors termination of pregnancy may be considered. The decision should be based on careful estimation of the entailed risk to the fetus, which in turn requires calculation of the dose to conceptus by a qualified expert. The decision itself should be made by the women to be treated in consultation with their physician, partner and counsellor. Particularly difficult problems arise when radiotherapy is performed in a woman with early, undiagnosed pregnancy. The result is sometimes a massive irradiation of the conceptus in a period when malformations are easily induced (at or after 3 weeks post conception). To avoid such unintentional irradiation it seems necessary to perform pregnancy tests to diagnose, or exclude the pregnancy before undertaking radiotherapy.
Therapy of hyperthyroidism with 131I in a pregnant woman is strictly contraindicated due to possibility of external irradiation of the foetus but mostly due to radioactive iodide crossing the placenta into the foetal circulation with subsequent uptake by its thyroid. The gland may well be destroyed by beta radiation from the nuclide taken up (131I). Therefore, other methods of treatment should be employed, if possible, until delivery.
When thyroid cancer with metastases is diagnosed in a pregnant woman, treatment with 131I, if it cannot be delayed after delivery, is not compatible with continuation of the pregnancy.
Can patients treatment with radiation endanger other people?
Medical radiation can be delivered to the patient from a radiation source outside the patient (e.g. from an x-ray machine for diagnosis or linear accelerator for radiotherapy). Regardless of how much dose the patient received, they do not become radioactive or emit radiation. As a result they present absolutely no radiation hazard to family or others.
The other way that medical radiation is given is by placing radioactive materials in the patient. In these cases the patient will emit radiation. For diagnostic nuclear medicine studies (such as a bone or thyroid scan) the amount of radioactivity injected is small and such patents present no hazard to their family or to the public. Such patients are discharged immediately after the procedure.
Patients may undergo radiation therapy by having radioactivity injected or radioactive sources implanted in the tumor. Such patients may or may not present a hazard to others based upon the penetration capability of the radiation emitted by the radionuclide. Some are very poorly penetrating (such as iodine-125 prostate implants. Such patents are discharged. Others who receive iridium-192 or cesium implants must remain in the hospital until the sources are removed.. The radiation in penetrating enough that visitors will be restricted from visiting the patient
Patients treated with high activity of 131I for cancer of the thyroid, in some cases for hyperthyroidism, or patients with permanent implants of radioactive sources (special category of brachytherapy), once released home from a clinic or hospital may present some - however slight - risk to their family members it they do not observe specific rules of behaviour in such situations. These patients must be informed orally to avoid close bodily contact with children and of other necessary precautions by specialists responsible for conduct of their therapy.
Table 1.
Deterministic effects after whole-body and localised irradiation by X and gamma rays; approximate absorbed threshold doses for single (short-term) and fractionated or low dose-rate (long-term) exposures [5,6].
Threshold absorbd dose Gy |
|||
| Organ/tissue | Effect | Short-term exposure (single doses) |
Long-term exposure (Yearly - repeated for many years) |
| Testicles | Temporal sterility permanent sterility |
0.15 3.5 - 6.0 |
0.4 2.0 |
| Ovaries | Sterility | 2.5 - 6.0 | > 0.2 |
| Ocular lens | Detectable opacities Visual impairment (cataract) |
0.5 - 2.0 5.0 |
> 0.1 > 0.15 |
| Bone marrow | Haemopoiesis impairment |
0.5 | > 0.4 |
| Skin | 1.Erythema (dry desquamation). 2. Moist desquamation. 3. Epidermal and deep skin necrosis 4. Skin atrophy with complications and telangiectasia |
2 18 25 10-12 |
- - - 1.0 |
| Whole body | Acute radiation sickness(mild) |
1.0 | - |
Table 2a.
Typical effective doses from diagnostic medical exposures in the 1990s (U.K.).
| Diagnostic procedure | Typical effective dose (mSv) |
| X-ray examinations: | |
| Limbs and joints
(except hip) Chest (single PA film) Skull Thoracic spine Lumbar spine Hip Pelvis Abdomen IVU Barium swallow Barium meal Barium follow thrugh Barium enema CT head CT chest CT abdomen or pelvis |
< 0.01 0.02 0.07 0.7 1.3 0.3 0.7 1.0 2.5 1.5 3 3 7 2.3 8 10 |
| Radionuclide studies: | |
Lung ventilation (Xe-133) |
0.3 |
Data from the National Radiation Protection Board in the U.K.
Table 2b. - alternative versions (from NRPB, modified).
| Broad levels of risk for common x-ray examinations and isotope scans | |||
| X-ray examination (or nuclear medicine isotope scan) | Effective doses (mSv) clustering around a value of: | Equivalent period of natural background radiation | Lifetime additional risk of cancer per examination* |
| Chest Teeth Arms and legs Hands and feet |
0.01 | A few days | Negligible risk |
| Skull Head Neck |
0.1 | A few weeks | Minimal risk 1 in 1 000 000 to 1 in 100 000 |
| Breast (mammography) Hip Spine Abdomen Pelvis CT scan of head (Lung isotope scan) (Kidney isotope scan) |
1.0 | A few months to a year | Very low risk 1 in 100 000 to 1 in 10 000 |
| Kidneys and bladder (IVU) Stomach - barium meal Colon - barium enema CT scan of abdomen (Bone isotope scan) |
10 | A few years | Low risk 1 in 10 000 to 1 in 1 000 |
| *These risk levels represent very small additions to the 1 in 3 chance we all have of getting cancer. | |||
Suggested information sources
Web sites for:
ICRP
NRPB
American College or Radiology
European Community
ASTRO
ESTRO
US National Cancer Institute
BMJ Evidence Based Medicine
ABSTRACT
The didactic text is devoted to patients' protection against unnecessary exposure to ionizing radiation.
There are obvious benefits to health from medical uses of radiation, i.e. in X ray diagnostics, in interventional radiology, nuclear medicine and radiotherapy. However, there are well-established risks from improperly applied high doses of radiation (therapy, interventional radiology) and possible deleterious effects from small radiation doses used in diagnostics. Appropriate use of large doses prevents serious harm from therapy but low doses carry a risk that cannot be entirely eliminated. Diagnostic use of radiation requires therefore such methodology that would secure high diagnostic gains while limiting the possible harm to the lowest possible level.
For assessment of the risk a quantitative measure of exposure is a necessary prerequisite. Dosimetric quantities are therefore explained and defined (absorbed dose, effective dose). Basic facts are presented on mechanisms of action of ionizing radiations on the living matter. Undesired deleterious effects in man are categorised into 1/ sequelae resulting from massive cell killing (the so called deterministic effects), requiring a high dose for their manifestation (exceeding the threshold dose), and 2/ into those effects originating from mutational changes in the cellular DNA, that may eventually lead to development of radiation induced cancer and of hereditary changes, transmitted to descendants of exposed individuals after irradiation of their gonads.
Data on the magnitude of threshold doses for cell killing effects are presented. On basis of experimental clinical and epidemiological evidence assessment is also given of the probability with which cancers and hereditary mutations may be induced by doses of various magnitude, most likely without a threshold dose (below which no effect would obtain).
The text provides ample information on opportunities to minimize the doses, and therefore the risk from diagnostic uses of radiation. This objective may be reached by avoiding unnecessary (unjustified) examinations, and optimizing the applied procedures both from the standpoint of diagnostic quality and of reduction of the excessive doses to patients. Optimization of patient protection in radiotherapy must depend on maintaining sufficiently high doses to irradiated tumours, securing high cure rate, while protecting the healthy tissues to the largest extent possible. Problems related to special protection of human embryo and foetus in course of diagnostic and therapeutic uses of radiation are presented and respective practical solutions are recommended.